|
Sumaiya Sayeed '20
0 Comments
by Gordon Wade '15 
Shark skin has so little drag that Speedo has made swimsuits with materials designed to imitate shark skin. [image via]
Evolutionary solutions to natural problems are often more effective than designed solutions. Biomimicry allows us to adapt these evolutionary concepts into great engineered designs. Sharks, for example, have evolved toward extremely high efficiency in traveling through water, meaning reductions in energy usage and increases in speed. Their physical skin design lowers drag forces associated with movement and has shown great potential for application in human transportation.
Sharks are peculiar in that they live in an aquatic environment and never stop moving. Because of this, they must find effective ways to transport themselves through fluids. One adaptation lies in the small-scale structure of their skin. Under a microscope, the surface of shark skin is seen to comprise thousands of small teeth-like structures called denticles. These denticles manipulate fluid dynamics in order to decrease drag. by Jennifer Maccani, PhD This article is part of the "Emerging Biotechnology" series. Is telepathic communication possible? As outlandish as it might sound, this question drove Hans Berger to investigate the electrochemical basis of brain activity—a line of research that eventually led to the invention of the electroencephalogram, or EEG, which measures the brain’s electrical impulses via electrodes attached to a person’s scalp (1). We can find clues as to what led Berger down this path by studying his early life. Berger was born in 1873 in Coburg, Germany. His diaries reveal that he was an introspective and solitary young man. After a short stint at the University of Berlin, Berger turned away from early leanings toward a career in astronomy and enlisted for military service in Würzburg, where a near-death experience radically altered his aspirations. One morning in 1892, Berger’s horse became spooked during an exercise with his artillery unit. Berger was thrown to the ground and into the path of an oncoming artillery cannon’s wheel. Although the cannon stopped just short of crushing him, Berger was thoroughly rattled. When his family sent him a telegram that evening inquiring about his wellbeing, they revealed that his sister had that very morning feared that something had happened to her brother. Berger became convinced that “it was a case of spontaneous telepathy in which at a time of mortal danger I transmitted my thoughts” (2). It may have been this very experience that led him, upon the completion of his military service, to attend Jena University in Jena, Germany and pursue his research on the electrical activity of the brain (2-4).  "In Germany I am not so famous." [image via] So... Why Make Glowing Animals? by Tiffany Citra '17 
Sherlock Holmes in his signature style--and also, let's take a moment to appreciate his adorable smile [left], with his companion "bachelor" John Watson [right]. [image via]
Some of you are probably already familiar with the “science of deduction,” a term made famous by the ongoing BBC TV show Sherlock. Along with his partner John Watson, wearing his signature deerstalker and overcoat, this modern-day Sherlock Holmes roams the bustling streets of London trying to solve all sorts of crimes, from the elementary ones to the so-called “three-patch problems.” And because it has had too many fangirls’ Tumblr posts already, we think it’d be cool to show our love for this amazing TV show the not-so-mainstream way: by discussing the science behind it.
Remember the “Hounds of the Baskerville”—the second episode of the second season, in which the 9-year-old Kirsty Stapleton filed a case about her “vanishing glow-in-the-dark rabbit” Bluebell? Bluebell is an example of genetic manipulation, which is the new kid on the block in the realm of biotechnology. We have been hearing about all sorts of benefits it may bring, from curing cancer to designing babies. The question, then, is why do some scientists spend their research, funding, and time to make animals… glow? by Sadhana Bala '17 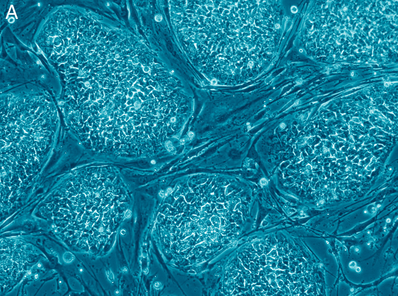 Human embryonic stem cells. [image via] In the United States, the biomedical significance of human embryonic stem cells (hESC) is recognized by Democrats and Republicans alike. Every passing month brings reports of the newest successful application of embryonic stem cells to a specific medical cause. hESC therapy has been used to further research on a wide variety of ailments – including spinal cord injury, multiple sclerosis, infertility and even hearing loss (1) – and it has generated largely promising results.
The controversy with hESC research does not center on the results but the methods – a moral dilemma that has been greatly debated in the media for years. Embryonic stem cells are derived from four- to five-day-old blastocysts, hollow balls of cells that represent the beginning stage of human embryo development. The extraction procedure often results in the destruction of a human embryo, usually one that has been voluntarily donated in a fertility clinic. But the huge potential of these cells has caused scientists, politicians, and the general public (2) to come to terms with this fact. Over the last two to three years, embryonic stem cells have, more or less, crawled off the agenda of the general media. Government-sponsored research for embryonic stem cells is currently at a pinnacle, yet it remains hindered due to one small piece of decade-old legislation by Jennifer Maccani, PhD This is part 3 of a 3-part article. This article is part of the "Emerging Biotechnology" series. It all began in 2008. That was the year Organovo Holdings, Inc. founder Dr. Gabor Forgacs of the University of Missouri–Columbia published an article in the journal Tissue Engineering Part A describing the first ever bioprinted blood vessel construct from chicken cells (15, 16). Rather than growing cells in a flat layer covered by cell culture media with the nutrients necessary for the cells’ survival, Dr. Forgacs and his colleagues had printed cells, or “bio-ink,” and collagen scaffolding (“bio-paper”) into a living three-dimensional structure (15). The beauty of his approach was that in a three-dimensional environment, the cells were free to do what their DNA programmed them to do—fuse with their neighbors and behave as they would in the body. This process of “self-assembly,” Dr. Forgacs explains, allows cells to retain their natural shape and characteristics that can sometimes be lost in the context of traditional two-dimensional cell culturing, such as in a petri dish (15). In the case of the chicken blood vessel constructs, the cells began to contract and “beat” (15)—just like a heart (16). If you’re still imagining the plight of the patient with heart failure, that’s good news—very good news. And it gets better.
by Jennifer Maccani, PhD This is part 2 of a 3-part article. This article is part of the "Emerging Biotechnology" series. A network of 3D printers is already beginning to form at Brown. One 3D printer is already in use in the laboratory of Dr. Kenneth Breuer, a fluid mechanist, and Dr. Sharon Swartz, a biologist focused on morphology and functional biology. Cosima Schunk, a graduate student in the lab, is a PhD candidate in the School of Engineering’s Biomedical Engineering Program just entering her fourth year. “My research is inspired by biomimetics, that’s my undergrad degree, and I’m looking at bat flight and possible application towards the development of engineering devices like micro-air vehicles,” Schunk says. One can tell from her undergraduate education at the University of Applied Sciences in Bremen, Germany, that Schunk is serious about the research of biomimetics; as she says, “It was the first school in Germany, actually, where you could directly study biomimetics.” As Schunk describes it, biomimetics is “generally learning from nature and applying biological concepts to engineering.” Although Schunk studies bats, “the idea, generally, is you look at bio-solutions evolved by nature and then you see if you can abstract the concept, and then apply this abstract concept towards an engineering solution.”  A bird emitting brightly colored chain links of goo. Or a model of how a bird's flapping wings create aerodynamic effects. [image via] In a recent publication in the Journal of Bioinspiration and Biomimetics (11), a former graduate student named Joseph Bahlman with whom Cosima Schunk has worked, together with Drs. Breuer and Swartz, characterized the robotic bat wing, or “flapper” as Schunk refers to it, that they designed and 3D-printed to study bat flight. “[Bats] are really agile flyers, very maneuverable, but we don’t know a lot about how they are actually doing it,” Schunk explains. “There’s a lot of engineering work out there looking at aeromechanical, aerodynamical behavior of membranous wings.”
by Jennifer Maccani, PhD This is part 1 of a 3-part article. This article is part of the "Emerging Biotechnology" series. Imagine being a patient with heart failure, waiting on the heart transplant list, knowing that a new heart is the only chance for your survival. Although you know that gaining your new life rests on someone else’s loss, you can’t help but hope that you might somehow be able to get to the top of the transplant list in time. Even if you do, there’s no guarantee that your body won’t reject the new heart, or that you’ll be able to tolerate potentially dangerous immunosuppressive drugs. Nearly 25% of heart transplant recipients suffer from organ rejection in the first year after transplantation (1). Now imagine that rather than waiting on the transplant list, your doctor could grow you a new heart and transplant it—without the harsh side effects of immunosuppressive drugs (2)—from your very own cells. That scenario may soon be a reality, thanks to a new technology called three-dimensional (3D) bioprinting. 3D bioprinting works like other types of 3D printing. Rather than depositing ink, 3D printers dispense materials such as plastic in consecutive layers according to a digital model to produce a three-dimensional structure (3). In this same vein, 3D bioprinters dispense spheres of cells—even, hopefully, a patient’s own cells—into a gel mold or scaffold to produce an organ or tissue (4). With bioprinting technology, tissues could potentially be used for transplants or for research. Furthermore, bioprinting has initiated a paradigm shift that could reduce the current reliance on animal models in studies of human diseases and in preclinical pharmaceutical drug testing (4). |

