|
by Jennifer Maccani, PhD This is part 3 of a 3-part article. This article is part of the "Emerging Biotechnology" series. It all began in 2008. That was the year Organovo Holdings, Inc. founder Dr. Gabor Forgacs of the University of Missouri–Columbia published an article in the journal Tissue Engineering Part A describing the first ever bioprinted blood vessel construct from chicken cells (15, 16). Rather than growing cells in a flat layer covered by cell culture media with the nutrients necessary for the cells’ survival, Dr. Forgacs and his colleagues had printed cells, or “bio-ink,” and collagen scaffolding (“bio-paper”) into a living three-dimensional structure (15). The beauty of his approach was that in a three-dimensional environment, the cells were free to do what their DNA programmed them to do—fuse with their neighbors and behave as they would in the body. This process of “self-assembly,” Dr. Forgacs explains, allows cells to retain their natural shape and characteristics that can sometimes be lost in the context of traditional two-dimensional cell culturing, such as in a petri dish (15). In the case of the chicken blood vessel constructs, the cells began to contract and “beat” (15)—just like a heart (16). If you’re still imagining the plight of the patient with heart failure, that’s good news—very good news. And it gets better. If that hadn’t gotten the attention of the medical community, Organovo’s current projects certainly have. Since Organovo developed the first commercial 3D bioprinter in 2009, the company has partnered with pharmaceutical companies such as Pfizer (17) as well as research institutions like Harvard Medical School (18), with the hope that Organovo’s 3D NovoGen™ MMX bioprinter (19) can be used in drug development and disease and regenerative medicine research. 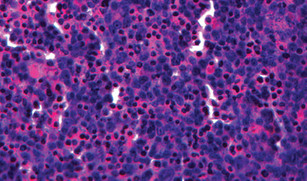 A cross-section of bioprinted liver. [image via] A cross-section of bioprinted liver. [image via] In April 2013, Organovo presented findings at the Experimental Biology conference in Boston, (19-21) describing a bioprinted liver model (21, 22). The liver model was made up of multiple cell types deposited in their natural architecture, capable of performing normal functions required of liver tissue—it produced proteins including albumin and fibrinogen, could be induced to secrete enzymes like the drug-metabolizing CYP1A2, and biosynthesized cholesterol. Basically, the model works just like liver tissue in the human body (21, 22). By demonstrating superior functionality, Organovo has set the stage for a revolutionary approach to testing new pharmaceuticals (20, 21), particularly in the wake of an article published in early 2013 in the Proceedings of the National Academy of Sciences by Dr. H. Shaw Warren and colleagues calling the accuracy and appropriateness of the use of animal models to research human diseases like sepsis into question (23, 24). As reported in The New York Times, independent medical experts have said that the article may change the course of such research (23). As an emerging technology with a myriad of potential clinical applications, 3D bioprinting is already improving the quality of care for dental patients and those in need of prosthetic limbs (25). Using 3D bioprinting, dental laboratories can take digital scans of patients’ teeth that can then be used to manufacture dental crowns, bridges, and implants, eliminating the need to take less accurate, uncomfortable molds while at the same time increasing affordability (25). In the field of prosthetics, 3D bioprinting has brought personalization and human dignity to patients needing prosthetic limbs, helping doctors to create leg prosthetics, for example, closely matching a patient’s remaining leg (25). However, there are hurdles the technology must overcome before it can advance into other applications. One potential issue with 3D bioprinting technology is long-term viability of bioprinted tissues. Without normal vasculature, cells lack the oxygen and other materials necessary for growth and differentiation—akin to suffocation (26). However, researchers at the University of Pennsylvania have published an article in Nature Materials last year that may solve that problem (27). Their approach consists of 3D-printed vascular templates made of sugar that dissolve once cells are added. Once the sugar mixture of sucrose, glucose, and dextran has flowed out through the channels left behind, fluid can flow through the bioprinted tissue. Bioprinting technology has the potential to improve clinical care and revolutionize human disease research, and may be a powerful tool for the advancement of both bedside medical practice as well as scientific pursuits at the bench. The future of 3D bioprinting and those who may benefit from it, not to mention the future of additive manufacturing in general, looks bright. As for Tyler Benster and his colleagues at Azavy? “In the fall we’re looking at a couple of different startup incubators including Betaspring… in the fall we would also start looking at raising a seed round of capital,” Benster says of his plans, in which he will work full-time at the company. If it’s successful, Azavy will help to connect designers and potential users to 3D printing resources, an endeavor that will have far-reaching benefits, even extending to ground-breaking medical and scientific advances. 3D printing has already had a positive impact on Cosima Schunk’s scientific research. “[3D printing] made the research as I’m doing it possible,” Schunk says. “We are scientists; we want to learn how a bat is flying and what we can learn from it, and that’s our major motivation.” When asked how 3D printing has allowed her to answer a research question she otherwise could not have, Schunk admits, “I could not think of an easy way how we would do it without a 3D printer.” In the end, the rise of 3D printing, especially its scientific and medical applications via 3D bioprinting, may sow a revolution-in-the-making within our own lifetimes—lifetimes that, if you’re a heart transplant patient, might be a lot longer, thanks to 3D bioprinting.
4 Comments
4/18/2022 04:40:10 am
Great article on 3D printing and its applications in Medical field. I have also written a article on how 3D printing industry can be used in Medical field. I hope you like it !
Reply
Hello
Reply
Thank you very much for your analysis. Our company is also engaged in related industries and your article is very helpful to us. Our company's website is https://zetarindustry.com
Reply
Leave a Reply. |
