|
By Olivia Woodford-Berry, '19 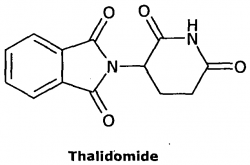 Though unfamiliar to some, many still remember thalidomide as one of the worst scientific mishaps of the twentieth century. This drug, originally produced in Germany, was brought to market with problematic lack of testing, and was later proven to cause an array of birth defects from phocomelia, or shortened or abnormal appendages resulting from problems with limb development in the womb, to autism like symptoms. (1) According to the Thalidomide Society, as many as 120,000 babies have miscarried, still born, or born with birth defects as a result of thalidomide. (1) This failure to regulate drugs has impacted thousands of victims, and the influence of this catastrophe continues to impact drug development in unseen ways. Specifically, the historical memories of thalidomide have given wake to the modern, virtually insurmountable power of the Food and Drug Administration. Thalidomide rolled out in several countries across Europe in the world, and while some doctors suspected a connection between thalidomide and specific incidences offside effects like nerve damage birth defects in their patients, Günenthal made every effort to conceal and deny these claims. While the drug was not approved in the U.S., this was not due to the scientific rigor at of the F.D.A. Even without approval, the drug circulated, going out to American doctors as “free samples” or as “trials” At this time, in fact, drugs that were not denied within sixty days were automatically approved. Rather, Francis Kelley, the particularly committed F.D.A. employee in charge of reviewing the application, suspected the inability of testers to find a lethal dose in rats suggested the rats could not absorb the drug in the ways that humans do— a suspicion that was later proven true. Despite pressures from the drug company and from within the F.D.A., she continued to declare the application incomplete, demanding that more testing be completed. This stubborn refusal on the part of a single employee is perhaps the only thing that kept the drug from being approved in the United States.
As the true consequences of thalidomide came to light, it began to disappear from the market. Still, the mechanisms of how thalidomide disrupted limb development remained unclear even as more and more victims were discovered. Thalidomide had been shown to disrupt several crucial processes from the regulation of growth factors to its inhibition of cell surface proteins. While several experiments suggested which features of thalidomide might play a role, there was no unifying pathway to explain the cases of phocomelia. Since thalidomide only caused problems in limb development and not the entire embryo, the mechanisms needed to have some sort of specifying feature that limited to these regions. It wasn’t until the year 2000 when one study suggested a unifying theory. Thalidomide’s chemical structure gave it a high binding affinity to the nucleotide guanine, one of the main components of our DNA. Genes encoding for cell surface proteins needed for formation of new blood vessels contain large sections of guanine in their promoter regions. Likewise, growth signaling molecules, like fGF-2, and also contain guanine heavy regions in their promoter region. Thus, thalidomide binds the promoter regions of these genes, preventing the expression of key genetic factors. This blocks formation of new blood vessels and the production of specific cell surface protein, two processes crucial for limb development. (4) While thalidomide was never approved in the United States, most see it as a fluke, and for those who regulate drug development, the lesson has been long lived. Today, the F.D.A. has more power than most other government agencies. Altogether, the F.D.A. regulates over 2.4 trillion dollars worth of drugs, and the scientists who consider applications of enormous pull. (5) Likewise, the burden of proof lies heavily on the drug makers to prove safety and efficacy, and all drugs must go through strict clinical trials before being mass distributed. In this way, the legacy of the thalidomide tragedy has shaped the present, virtually all powerful leash on drug makers today. Sources:
0 Comments
Leave a Reply. |
