|
By Olivia Woodford-Berry, '19 While science is often viewed as an objective, well-policed corner of academia, the trust in the integrity of scientists is not always well placed. Certainly, not all “bad science” qualifies as fraud, but gray areas such as misrepresentations of results, research bias, and formation of hypothesis to fit pre-gathered data are pervasive in research, as one third of scientists admit to participating in some “questionable practices” in their carriers. [1] Misconduct is often driven by a desperation to believe that a meaningful hypothesis is correct, and researchers, consciously or unconsciously, may see their data through rose colored glasses. Furthermore, studies have shown that scientific misconduct is on the rise, and perhaps no sub-field has experienced this reality more than stem cell biology. [2] 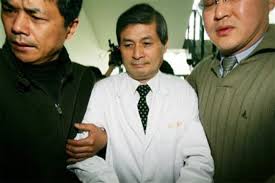 Stem cell biology quickly became a hot field with promising applications in regenerative medicine. Predictably, a sort of race emerged among scientists to find the next new population of stem cells. Such a discovery promised grants, prestige, and perhaps even consideration for a Nobel Prize; Hence, misconduct began to rear up amongst those determined to be winners in the field. In 2006, a committee investigating the laboratory of Woo Suk Hwang at Seoul National University determined they fabricated data in two studies which claimed to clone human embryos. [3] While this served as a devastating blow to the university, once acclaimed for housing South Korea’s “supreme scientist,” this lesson was not as far reaching as the scientific community would have hoped, and similar accusations were recently brought to light in leading research institutions in the United States. Dr. Piero Anversa’s research career truly took off at an American Heart Association meeting in the year 2000 when he broadcasted the groundbreaking claim that injecting bone marrow stem cells into mouse hearts induced tissue regeneration. While controversial at the time, the discovery set researchers across the field into a frenzy. He went further to claim the discovery of endogenous heart stem cells that had therapeutic utility. Despite growing numbers of respected labs failing to reproduce data supporting either of those claims, Harvard took years to complete investigations debunking his fraudulent experiments. [4] [5] Only this year, Harvard accuse the prominent cardiac researcher of fabricating data in over 30 published studies. While Anversa admits that collaborators manipulated data in a few of the cases of interest, he claims such behavior was without his knowledge and that the broader conclusions of his career still hold scientific weight. [6] Still, while he seemed to grow outstanding levels of new tissues, others have seen nothing close to his supposed data. Clinical trials and companies based on cardiac regeneration still exist, though such trials have since been paused. While there are surely bad scientists to blame, there are often systematic factors at play from universities emphasizing the need to attract prestigious grants and the high cost of whistle-blowing on peers in one’s field. Likewise, in the face of multiple possible interpretations, clinicians and scientist alike cling to any reason to maintain hope that their research will be the next breakthrough. Going forward, the scientific community faces new questions about how to maintain scientific rigor without shutting out new ideas.
Sources:
1 Comment
|

