|
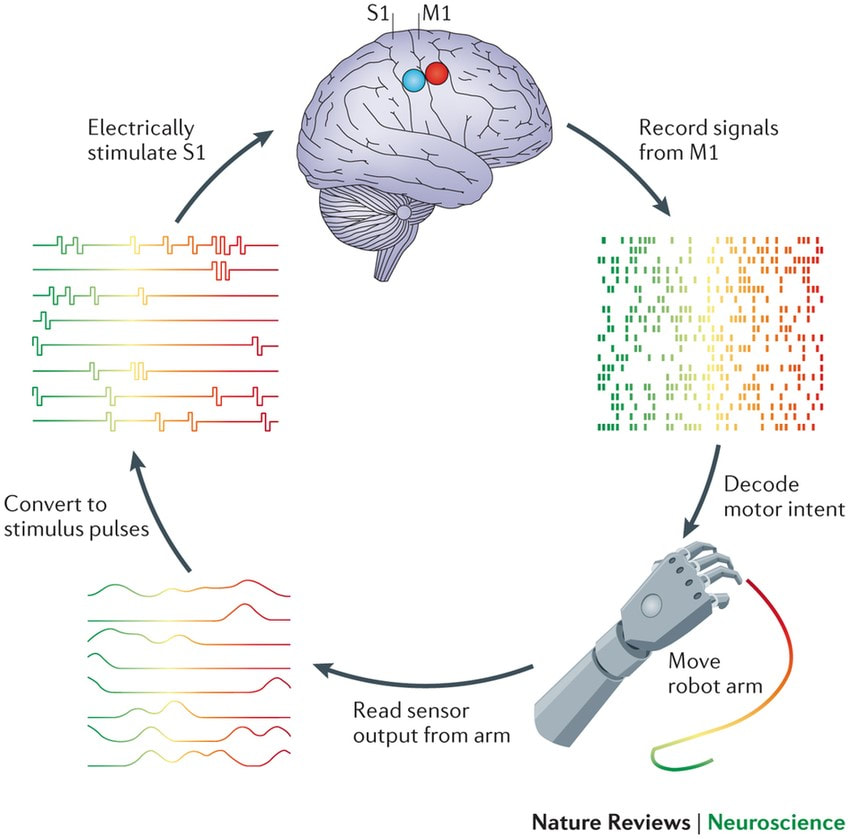
By Jess Sevetson Figure courtesy of Sliman J Bensmaia & Lee E Miller. “Restoring sensorimotor function through intracortical interfaces: progress and looming challenges” Nature Reviews Neuroscience 2014 May The term “mind reading” usually suggests the ability to read out someone’s thoughts like an audiobook. While there has been some progress on that front recently, another application of these technologies is the ability to operate advanced prostheses. Recording from areas of the brain that deal with motor information allows scientists to develop robotic limbs that respond to the brain’s commands. Similarly, cochlear implants function by stimulating areas of the brain involved in hearing. Traditionally, direct recording and stimulating meant piercing brain tissue with one or many metal electrodes, known as a Multi-Electrode Array (MEA). Over time, however, this leads to damaged, scarred tissue, which then makes recording activity difficult [2]. A solution exists in the form of Electrocorticography (ECoG) grids, which sit on the outer surface of the brain rather than penetrating it. Traditional ECoG grids still lack some spatial acuity. This is due in part to their distance from the deeper layers of neural tissue, as well as the large size of their electrodes. Given these difficulties in recording electrical signals in the brain, a recent development by Konerding and colleagues offers the potential for improved precision. Their paper, published in Nature this past month, showed consistent mapping of guinea pig auditory cortex at an improved resolution. The new thin-film surface electrode arrays allow researchers to pick up two important neuronal components: local field potentials and multi-unit activity. Traditional ECoG units record from areas at least 1 millimeter in diameter, which may sound small but actually encompasses the activity of thousands of cells. These measurements, known as local field potentials, can be used to see what a region of cells are doing, but can’t tell us much about the individual neurons. Conversely, MEAs are used to pick up what’s known as multi-unit activity, or a summation of a handful of cells which can then be broken down into their individual contributions. Through advances in electrode array design, Konerding and colleagues offer a more exact spatial area, as well as the ability to pick up on both of these types of activity. While these advancements may have improved brain recording, there are still drawbacks in using ECoG grids. One such drawback intrinsic to ECoG recordings is that they are most successful at recording the activity of superficial neurons, and cannot reliably record from deeper areas of the brain. Additionally, the end goal of Brain-Machine Interfaces is to not only listen to the activity of the brain, but to also actively communicate with it, such as with cochlear implants. More research is required to determine whether this new device will also provide similar advances in allowing us to stimulate the brain tissue. Despite these caveats, optimized ECoG grids unquestionably enhance our ability to understand the activity of the brain. Improved spatial resolution and paired local field potential and multi-unit recordings are a step forward on the road to more elegant prostheses. [1] W. S. Konerding, U. P. Froriep, A. Kral & P. Baumhoff. “New thin-film surface electrode array enables brain mapping with high spatial acuity in rodents” Nature, Feb 2018
[2] Vadim S. Polikov, Patrick A. Tresco, William M. Reichert. "Response of brain tissue to chronically implanted neural electrodes" Journal of Neuroscience Methods, 2005
0 Comments
Leave a Reply. |